| GENERAL DESCRIPTION |
 In addition to the normal requirements for a good broadcast transmitter such as high fidelity, low operating cost, and easy maintenance, the good shortwave transmitter must possess certain other requisites. Most important among these is that frequency of operation be changed as many as six times a day with a minimum of off-air time. The transmitter must also operate into an antenna, or antenna system, having standing-wave ratios as high as 1.5:1 and impedances from 300 to 600 ohms.
In addition to the normal requirements for a good broadcast transmitter such as high fidelity, low operating cost, and easy maintenance, the good shortwave transmitter must possess certain other requisites. Most important among these is that frequency of operation be changed as many as six times a day with a minimum of off-air time. The transmitter must also operate into an antenna, or antenna system, having standing-wave ratios as high as 1.5:1 and impedances from 300 to 600 ohms.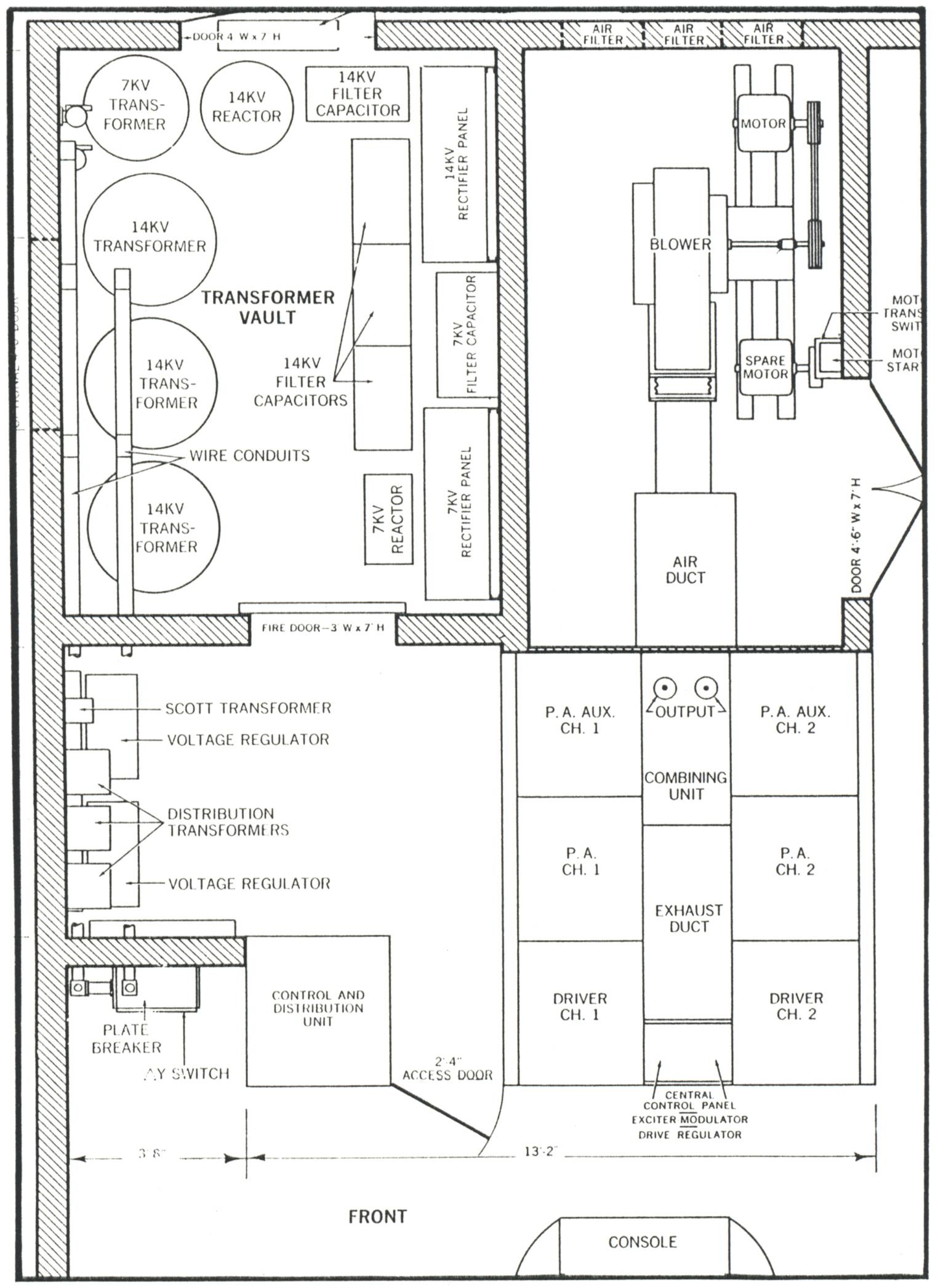 Ampliphase Features
Ampliphase Features Special Electrical-Mechanical Component Design
Special Electrical-Mechanical Component Design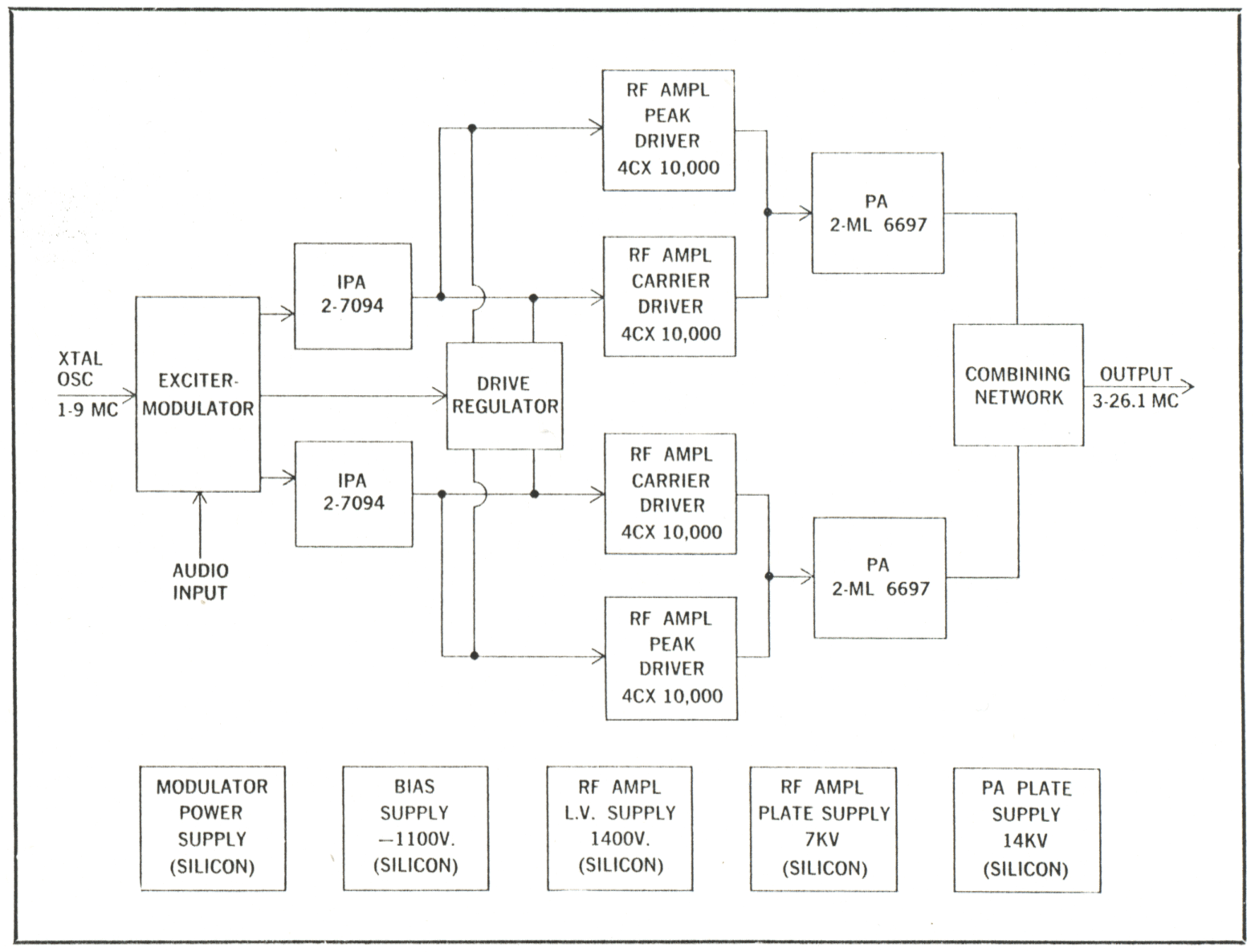
| TUBE COMPLEMENT | |||||
| RF stages | AF stages and modulator | Rectifiers | |||
| Number | Type | Number | Type | Number | Type |
| 4 | ML-6697 | ||||
| 4 | 4CX10,000 | ||||
| 4 | 7094 | ||||
| THIS TYPE OF TRANSMITTER IS INSTALLED IN THE FOLLOWING COUNTRIES | |||||
| ITU | Country | ITU | Country | ||